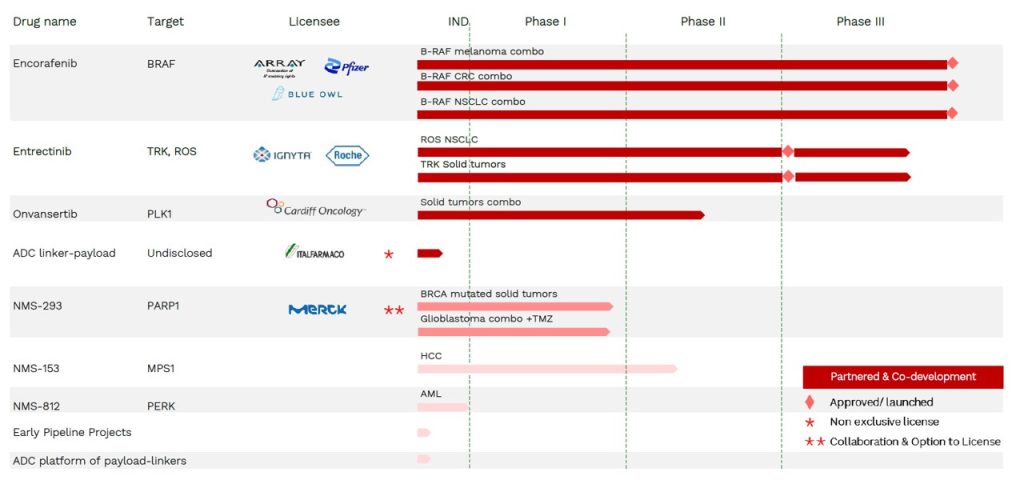
Pipeline
 Encorafenib
Encorafenib
Encorafenib is a small molecule inhibitor of the BRAF kinase, a key enzyme in the tyrosine kinase/MAPK signaling pathway (TK-RAS-RAF-MEK-ERK), which is often mutated and activated in different tumors, such as melanoma, colorectal cancer, non-small cell lung cancer and others. Encorafenib is a potent BRAF inhibitor for which NMS has licensed enabling IP rights for commercialization. BRAFTOVI® (the brand name for encorafenib) is approved in combination with the MEK inhibitor binimetinib (MEKTOVI®) for the treatment of unresectable or metastatic melanoma with a BRAFV600E or BRAFV600K mutation. It is also the first BRAF inhibitor approved in combination with cetuximab (Erbitux®) for the treatment of adult patients with metastatic colorectal cancer (CRC) with a BRAF V600E mutation. More recently, the FDA and EMA have approved BRAFTOVI® + MEKTOVI® for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) with a BRAF V600E mutation. Royalty Agreement signed with Blue Owl Capital. Deal announced on 5 September 2024.
 Entrectinib
Entrectinib
Entrectinib is a selective inhibitor of the tyrosine kinases TRK A/B/C, ROS1 and ALK, whose activating gene rearrangements drive proliferation in small subsets of different tumor types. Entrectinib was invented and developed into Phase I by NMS, then licensed to Ignyta (San Diego, USA) and subsequently acquired by Roche. Entrectinib is one of the first drugs approved with an agnostic indication for adult and paediatric patients with relapsed/refractory tumors harbouring NTRK gene fusions, independently form the tumor type. It is also approved for the treatment of adults with ROS1-positive, metastatic non-small cell lung cancer (NSCLC). Entrectinib was specifically designed to efficiently penetrate the blood brain barrier and it is efficacious also in patients with brain metastases, with a good tolerability profile.
 Onvansertib
Onvansertib
Onvansertib is the first orally available, potent and selective inhibitor of the PLK1 kinase, a master regulator of mitotic progression which is overexpressed and activated in proliferating cancer cells. The drug was invented and developed into Phase I by NMS, then licensed to the US Biotech Trovagene/Cardiff Oncology. Clinical development is currently ongoing with promising results in Phase I/II studies in combination, including a Phase Ib/II study with FOLFIRI/bevacizumab for treatment of metastatic KRAS mut CRC. For a comprehensive, up-to-date, Onvansertib overview please refer to Cardiff Oncology web site.
 NMS-153
NMS-153
NMS-153 is a potent, highly selective inhibitor of MPS1, with potential for first-in-class. MPS1 is a kinase found to be highly expressed in a number of human tumors that plays a critical role in the control of mitosis by regulating the spindle assembly checkpoint, a mitotic mechanism required for proper chromosome alignment and segregation during cellular division. NMS-153 is a molecule with long residence time on the target and selective against a wide range of tested enzymes. In vitro, a brief exposure to the compound is sufficient to commit cancer cells to death. Its anti-proliferative activity is associated with a demonstrated mechanism of action in different tumor cell lines, including hepatocellular carcinoma and broad in vivo efficacy both as single agent and in combination with standards of care. NMS-153 is currently in Phase I/II clinical development in hepatocellular carcinoma as single agent (NCT05630937) and combination studies are planned.
 NMS-293
NMS-293
NMS-293 is a second generation PARP inhibitor that differentiates from other approved or advanced molecules in its unique selectivity for PARP1 vs PARP2 enzymes and low DNA trapping activity, both features potentially linked to lower haematological toxicity and higher potential for combination with DNA damaging agents in a wide range of tumors, covering high unmet medical needs. It also has a superior ability to penetrate the blood brain barrier, a very important feature supporting its utilization in CNS tumors and brain metastases. The drug has shown high anti-tumor activity as single agent in BRCA mutated preclinical tumor models and synergy and tolerability in combination with chemotherapy.
Based on these findings, NMS-293 is currently in clinical development as monotherapy for the treatment of patients with BRCA mutated breast, ovarian, prostate and pancreatic tumors (NCT04182516) and in combination with temozolomide in recurrent glioblastoma (NCT04910022).
 NMS-812
NMS-812
NMS-812 is a novel, potent and orally bioavailable dual inhibitor of PERK (PKR-like endoplasmic reticulum kinase) and GCN2 (General Control Nonderepressible 2), with potential for first-in-class in several potential oncology indications. PERK and GCN2 are effectors of the Integrated Stress Response (ISR), a pro-survival pathway exploited by cancer cells to survive stress. The dual inhibition of the ISR, the pathway involved in survival of cancer cells to standard therapy may potentially overcome drug resistance and offer superior anti-proliferative activity. In addition, NMS-812 also modulates the immune response via direct and indirect mechanisms which may contribute to anti-cancer activity. The first in human (FIH) study showed excellent pharmacokinetic profile allowing daily oral dosing and likely permissive safety for further development. Based on preclinical data and its unique, dual inhibition of the two key components of the Integrated Stress Response mechanism, NMS-812 may represent a novel strategy for Acute Myeloid Leukemia (AML), with potential for synergies with other drugs and potential to overcome drug resistance.

